
Innovative Scarless Ultrasound-Guided Treatment for Benign Neck Tumors
Department of Otolaryngology, Far Eastern Memorial Hospital
Director: Dr. Li-Jen Liao
Head and neck soft tissue ultrasound is the first-line tool for evaluating neck masses. Since 2007, the Department of Otolaryngology at Far Eastern Memorial Hospital has provided head and neck ultrasound services, with a history of over 16 years. Each year, approximately 2,000 to 3,000 patients undergo examination. In addition to diagnostic use, we have developed ultrasound-guided treatments in recent years to help manage benign neck tumors. Our department has become one of the leading centers in Taiwan for head and neck ultrasound examinations.
Over the past 30 years, soft tissue ultrasound has been widely used to evaluate head and neck lesions, including thyroid enlargement, nodules, lymph nodes, salivary gland tumors, and other masses. It can also assess neck vessels, vocal cord mobility, and major joints. When abnormalities are detected, physicians may recommend ultrasound-guided fine needle aspiration or core needle biopsy for early diagnosis and timely treatment.
Ultrasound-Guided Ethanol Injection
Ultrasound can assist in localization before surgery and precisely identify lesions during procedures, especially for non-palpable masses. Preoperative contrast injection and ultrasound marking can significantly improve surgical success rates.For benign cystic lesions of the neck, such as thyroid cysts, ultrasound-guided aspiration is performed to remove fluid and reduce swelling, followed by cytological examination. In cases of recurrent swelling, treatment options include surgical excision or ultrasound-guided injection of sclerosing agents. Our hospital utilizes 99.5% medical-grade ethanol for sclerotherapy. The procedure is similar to cyst drainage, with a small amount of ethanol injected at the end. Patients do not experience intoxication, although mild swelling or pain may occur for 1–2 days. Over 80% of cysts improve after a single treatment. This procedure was approved for National Health Insurance coverage in December 2021 for recurrent benign thyroid cysts. For other benign cysts not covered, self-paid treatment has been approved by the New Taipei City Health Bureau. This work received the First Prize for Outstanding Ultrasound Research from the Taiwan Society of Ultrasound in Medicine in 2021.
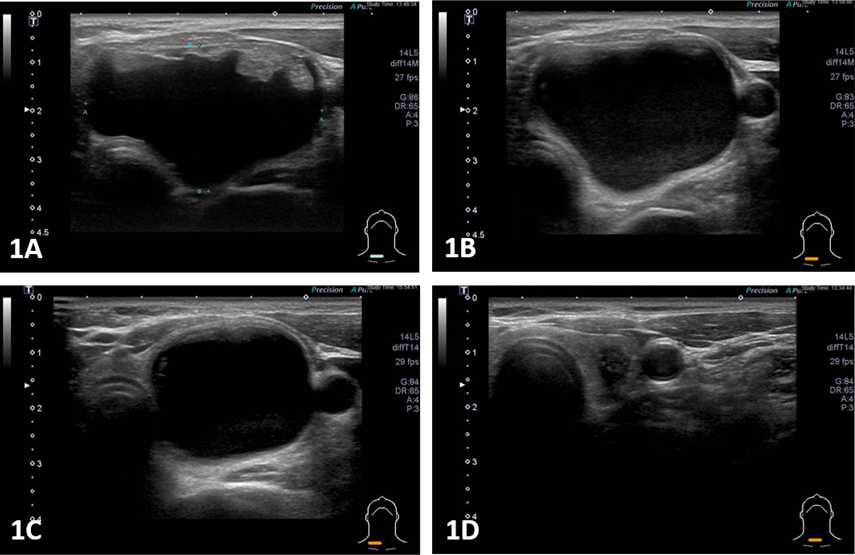
Figure 1. Effect of ultrasound-guided ethanol injection
1A: Before injection; 1B: 1-month follow-up; 1C: 3-month follow-up; 1D: 6-month follow-up
Ultrasound-Guided Ablation Therapy
While ethanol injection is suitable for recurrent cystic lesions, it is less effective for solid benign thyroid nodules. These nodules may cause compression symptoms or cosmetic concerns. Traditional surgery or endoscopic surgery may leave scars, whereas ultrasound-guided ablation provides a scarless alternative. Under ultrasound guidance, an ablation needle is used to treat the tumor. This therapy is suitable for nodules around 4 cm, typically reducing to less than 2 cm after six months, achieving satisfactory cosmetic outcomes.
It should be noted that both ethanol injection and ablation reduce tumor size rather than completely eliminate it. The main advantage is the absence of scarring. However, post-treatment ultrasound images may resemble abnormal lesions, so regular follow-up with the same physician is recommended.
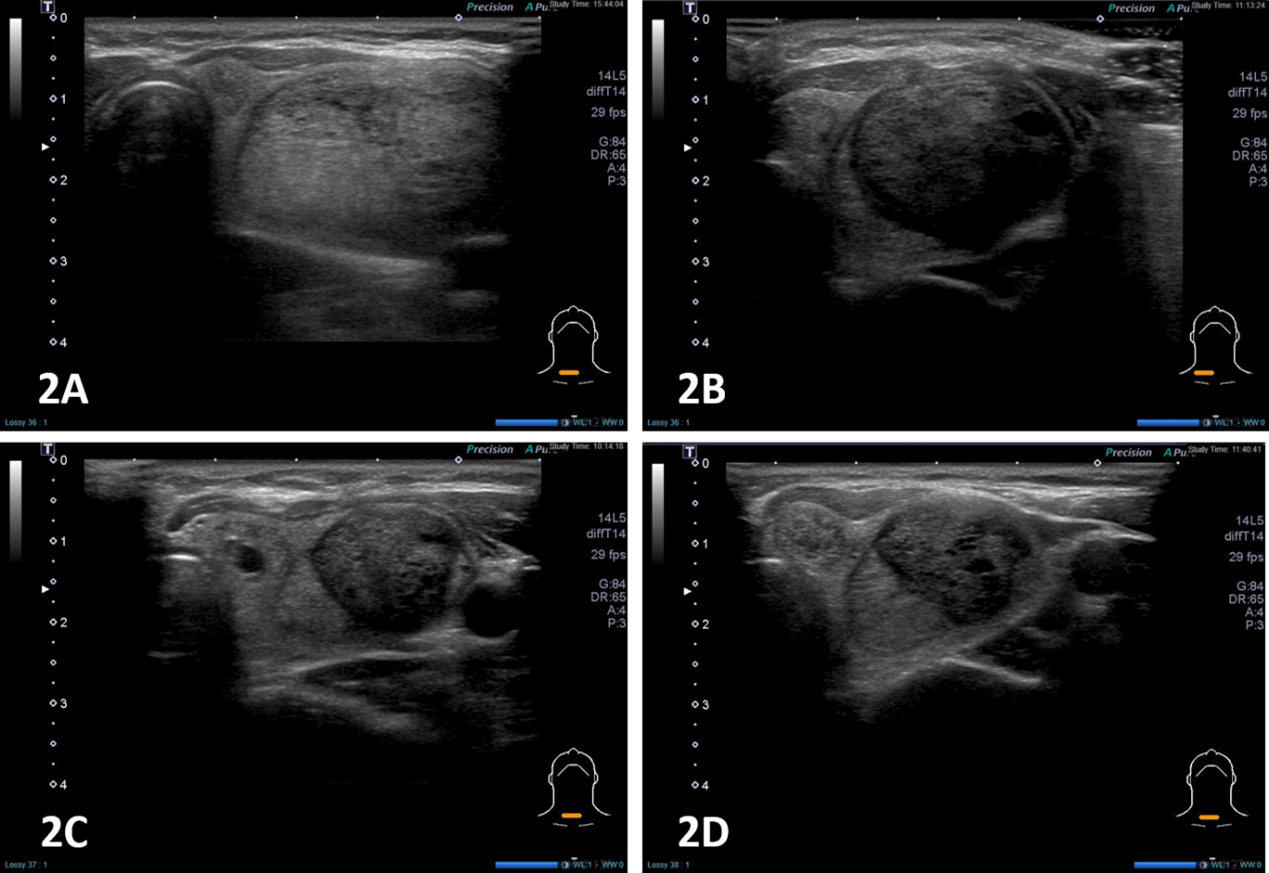
Figure 2. Effect of ultrasound-guided ablation therapy
1A: Before ablation; 1B: 1-month follow-up; 1C: 3-month follow-up; 1D: 6-month follow-up
Non-invasive High-Intensity Focused Ultrasound (HIFU)
In 2024, the Department of Otolaryngology plans to introduce the latest non-invasive High-Intensity Focused Ultrasound (HIFU) technology (Figure 3). This method does not require needle insertion; instead, ultrasound energy is directly focused on the lesion to achieve ablation. The application is currently under review by the New Taipei City Health Bureau, and we hope to introduce this technology in 2024 to benefit more patients.
Figure 3. Planned introduction of HIFU technology in 2024.
